Instructor(s) name(s) and contact information
Chloe Mirzayi, cmirzayi@gmail.com
Levi Waldron, levi.waldron@sph.cuny.edu
CUNY School of Public Health, 55 W 125th St, New York, NY 111027
Introduction: Just what is a cause anyway?
Question: In your own words, what is a cause? What does it mean to cause something?
How do we know our potential cause brought about an effect or a result? Perhaps it preceded the effect of interest. However, it could still just be a coincidence. Perhaps there is a lurking variable that truly causes both what we think is the cause and the observed effect.
Despite this, humans (and animals) tend to have a relatively intuitive understanding of causation when we observe it directly. When I burn my finger while cooking, I know what caused it–my finger coming into contact with a hot stove. I could also trace the causal pathway backward and identify what caused my finger to come into contact with the hot stove in the first place–such as my motivation to cook dinner or me being distracted by the antics of my cat.
Example: My cat, who I adopted in 2017, has quickly learned that jumping on my kitchen table while I’m eating results in him getting a spritz of water with a spray bottle–but only if the spray bottle is within reach. What potential causal variables could my cat be considering when he decides to jump on the table?
The counterfactual definition of a cause
However, the nature of much of the work I (and probably most of you) do does not allow us to directly observe what caused an event of interest. Instead we must rely on inferences we draw from our data to establish an argument for a particular causal mechanism or pathway.
Rather than relying on vague dictionary definitions or me talking at length about my cat to inform the science of causal inference, the most commonly accepted definition of a cause in modern epidemiology is based on a counterfactual: what would have happened had the event of interest had the exposure not occurred.
But that’s not very intuitive or easy to parse so let’s consider an example:
Example: A dog and an ambulance
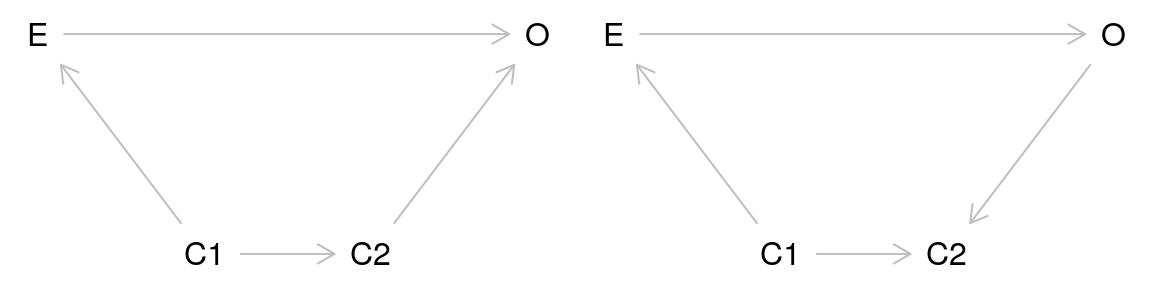
An ambulance drives by a house with its sirens on. A dog in the house barks. We can ask the causal question in a straightforward way: Did the ambulance driving by the house cause the dog to bark?
Alternatively, we can rephrase the question in terms of a counterfactual: Would the dog have barked if the ambulance had not driven by the house? We can visualize the relationship using a causal diagram also called a directed acyclic graph (DAG).

This diagram depicts the cause or exposure (the ambulance) and the effect or the outcome (the dog barking) with an arrow indicating the direction of the causal relationship. In contrast to a diagram showing a statistical relationship, causal diagrams must state a directional relationship because we are theorizing that one variable causes another.
This is the simplest form of a causal diagram. If we were to statistically model the effect for this relationship using regression, a simple two-variable model containing the exposure and the outcome would give us a correct, unbiased estimate of the actual strength of the cause on the effect. However, we are rarely that lucky. A common source of bias is confounding.
Question: Can you think of any situations that may bias the observed relationship between the ambulance and the dog barking?
Bias and Confounding
Confounding
Confounding arises when a third variable is present in the relationship between cause-and-effect. A confounder is present when:
- The confounder causes the outcome
- The confounder causes the exposure
- The confounder is not a mediator (i.e. it is not present on the causal pathway between the exposure and the outcome)
Returning to our example, the relationship between the ambulance and the barking dog could be confounded by a third variable: a nearby car crash. Perhaps this car crash is what started the dog barking before the ambulance even arrived, but the ambulance caused the dog to bark even more.
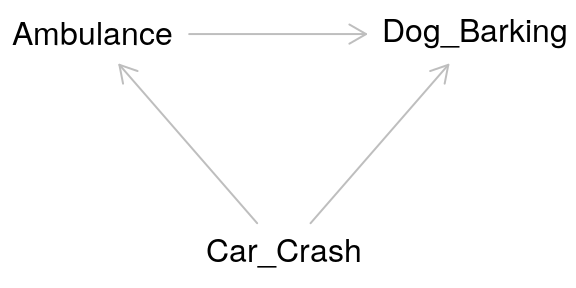 The pathway between the exposure and the outcome through the confounder is called a “backdoor pathway” and it can bias the relationship between the exposure and the outcome you observe. In this situation, we might observe barking and conclude that it occurred due to the ambulance when in fact it was due to the car crash. Or we might conclude that the volume of the barking caused by the ambulance alone is much greater than it actually is due to the combined effect of both the car crash and the ambulance.
The pathway between the exposure and the outcome through the confounder is called a “backdoor pathway” and it can bias the relationship between the exposure and the outcome you observe. In this situation, we might observe barking and conclude that it occurred due to the ambulance when in fact it was due to the car crash. Or we might conclude that the volume of the barking caused by the ambulance alone is much greater than it actually is due to the combined effect of both the car crash and the ambulance.
We can “deconfound” the effect of interest by controlling for the confounder. The old-fashioned way of doing this is to manually stratify your data by levels of the confounder. Then you calculate an effect size for each stratum. In a modern regression model, we can include the confounder as an independent variable in our model and it is controlled for. When we include the confounder as a variable in a regression model, we are effectively stratifying the effect across different levels of the confounder then taking the weighted average across strata to get an effect size.
Colliders
Taken at face value, colliders are similar to confounders. However, colliders are not common causes of the exposure and outcome. Instead:
- The outcome causes the collider
- The exposure causes the collider
- The collider is not a mediator
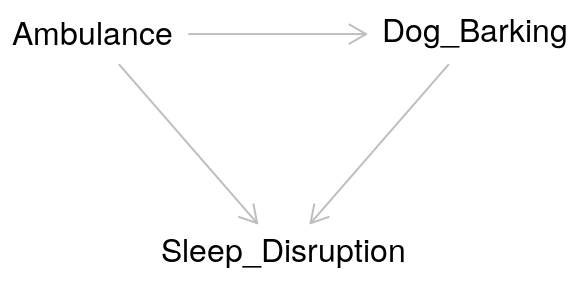
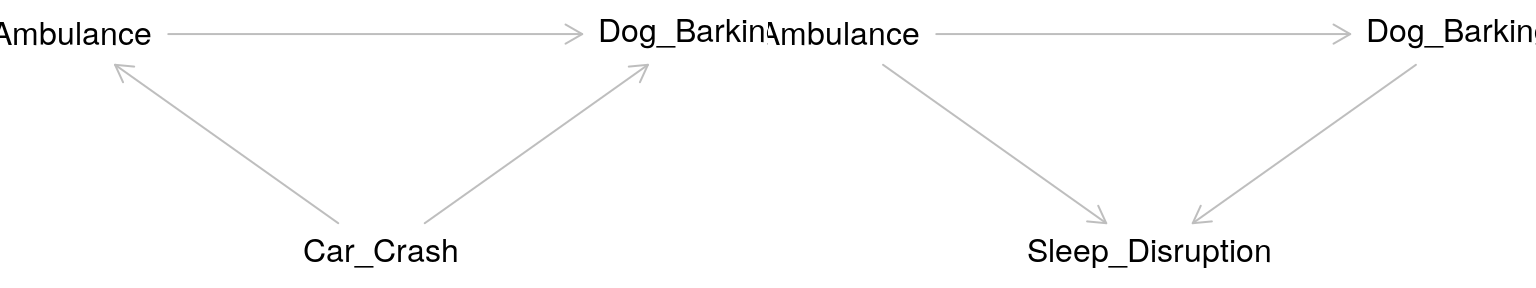
Adjusting for a collider as one would for a confounder can create bias. In effect by adjusting for a collider, a backdoor pathway is opened between the exposure and the outcome through the collider. So what’s the proper way of dealing with colliders? Ignoring them! Hypothesized colliders should not be adjusted for or included in models.
Selection bias
Another major issue of causal inference is selection bias. This arises when selection is dependent on the exposure and the outcome in the study:
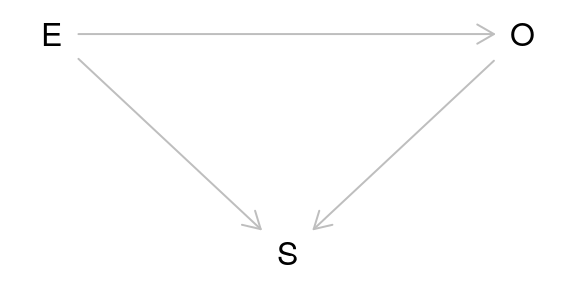
As can be seen from the DAG above, selection bias is a collider. In this case, the study is conditioned on the collider by the act of selection for the study. As a result, the study is biased. One particularly memorable way to think of selection bias is as the “already dead” problem. People who have already died of the outcome are not alive to be in the study. These people may have a more aggressive or serious form of the outcome and not including them masks some of the causal relationship.
A common source of selection bias is loss to follow-up in many studies. In survival analysis in particular this can be important as the people who are lost may differ in important ways from those who completed the study. Unfortunately because these people were lost, it is often difficult to assess how they differed from those who were not lost.
Study Designs
Randomized Control Trials (RCTs)
- Exposure of interest is assigned at random
- Because exposure is assigned at random, there are no possible confounders of the relationship between exposure and outcome
- Considered the gold standard for causal inference
- Selection bias is still possible
Question: How can selection bias occur in an RCT?
Instrumental Variable Analysis/Mendelian Randomization
- Attempts to mimic an RCT using a source of pseudorandomization
Consider the DAG for an RCT:

In IV/MR analysis the coin flip is replaced with a source of randomization such as genetic variation: 
Researchers have used a variety of factors as sources of pseudorandomness in IV studies: - Before and after implementation of a new government policy - Distance from a medical center -
Cohort Studies
- Participants are chosen on a common characteristic (such as occupation, location, or history of a particular disease)
- They are then followed over time and exposures and outcomes can be assessed
- Selection bias and confounding can occur
Causal Inference in R
R provides many packages that are helpful in causal inference.
Dagitty
Dagitty allows for creating causal diagrams, but also gives you more information including what variables to adjust for in complicated causal models.
g1 <- dagitty( "dag { W1 -> Z1 -> X -> Y Z1 <- V -> Z2 W2 -> Z2 -> Y X <- W1 -> W2 -> Y }") plot(graphLayout(g1))

for( n in names(g1) ){ for( m in setdiff( descendants( g1, n ), n ) ){ a <- adjustmentSets( g1, n, m ) if( length(a) > 0 ){ cat("The total effect of ",n," on ",m, " is identifiable controlling for:\n",sep="") print( a, prefix=" * " ) } } }
## The total effect of V on Z2 is identifiable controlling for:
## * {}
## The total effect of V on Y is identifiable controlling for:
## * {}
## The total effect of V on Z1 is identifiable controlling for:
## * {}
## The total effect of V on X is identifiable controlling for:
## * {}
## The total effect of W1 on Z1 is identifiable controlling for:
## * {}
## The total effect of W1 on X is identifiable controlling for:
## * {}
## The total effect of W1 on Y is identifiable controlling for:
## * {}
## The total effect of W1 on W2 is identifiable controlling for:
## * {}
## The total effect of W1 on Z2 is identifiable controlling for:
## * {}
## The total effect of W2 on Z2 is identifiable controlling for:
## * {}
## The total effect of W2 on Y is identifiable controlling for:
## { V, X }
## { W1 }
## The total effect of X on Y is identifiable controlling for:
## { W2, Z2 }
## { V, W2 }
## { V, W1 }
## { W1, Z1 }
## The total effect of Z1 on X is identifiable controlling for:
## { W1 }
## The total effect of Z1 on Y is identifiable controlling for:
## { W1, W2, Z2 }
## { V, W1 }
## The total effect of Z2 on Y is identifiable controlling for:
## { W2, X }
## { W1, W2, Z1 }
## { V, W2 }ggdag
Another option for making causal diagrams is ggdag which can accept daggity objects and plots DAGs using ggplot2.
ggdag_adjustment_set(g1, "X", "Y")

DESeq2
DESeq2 uses negative binomial regression. As such, it allows you to deconfound by including the confounder as a variable in the design statement:
ddsSE <- DESeqDataSet(se, design = ~ exposure + confounder1 + confounder2) ddsSE
edgeR and limma
You can use model.matrix() to specify the exposure and confounders:
batch <- factor(ds$Batch) treat <- factor(ds$tx) y <- factor(ds$surv) design <- model.matrix(~treat + batch)
Then fit the model either in edgeR:
fit <- glmQLFit(y, design)
Or in limma:
fit <- lmFit(y, design)
As mentioned in the confounding section, estimates obtained for the relationship between the exposure and the outcome have been adjusted for the included confounders (deconfounded). This means that the observed estimate is an averaged effect size across strata of the confounder(s).
Example in curatedMetagenomicData
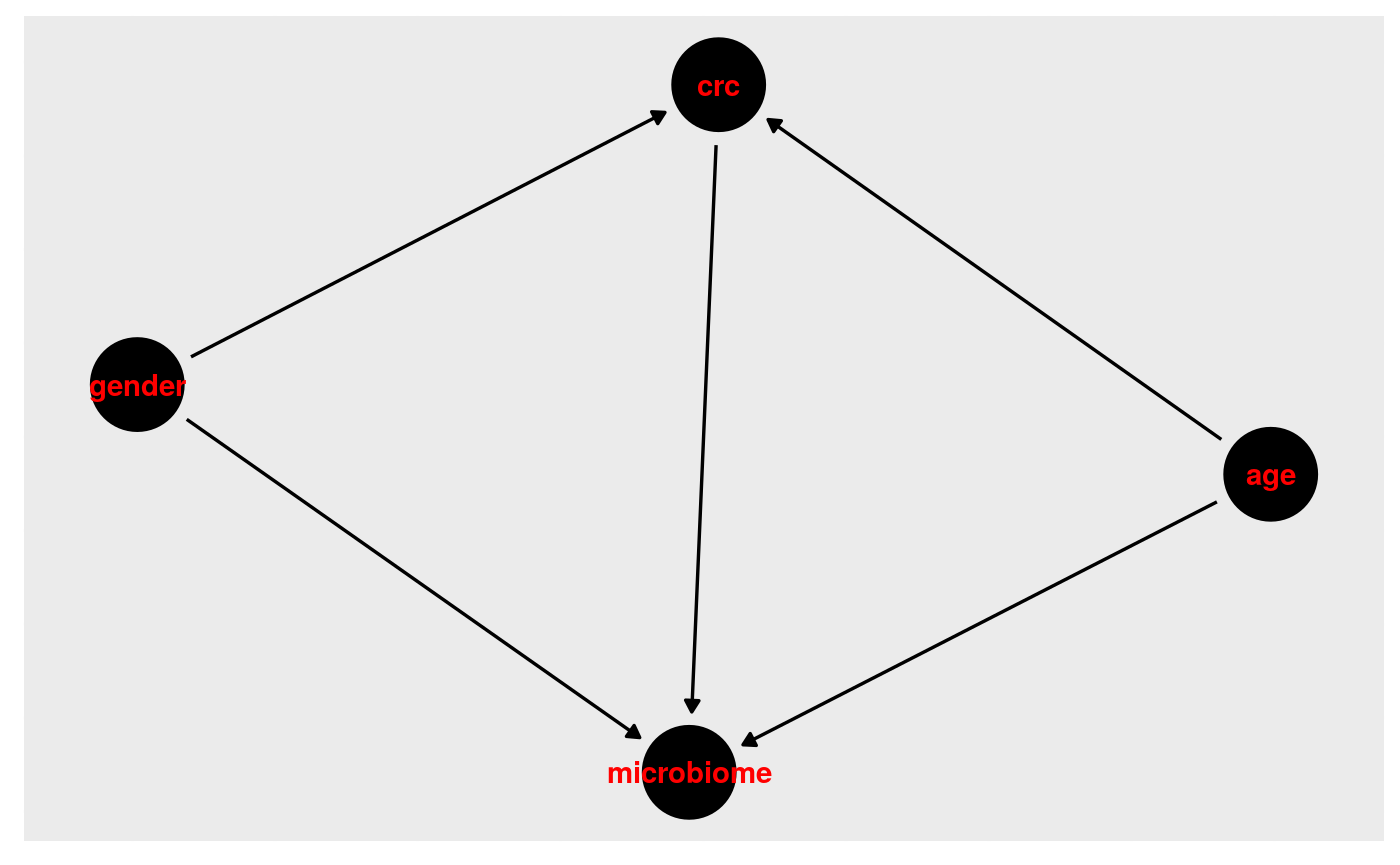
Step 1: Draw our assumptions. For this example, we use the VogtmannE 2016 dataset in cMD. The dataset has 52 cases of colorectal cancer and 52 controls. First, let’s create a causal diagram for this relationship.
v_dag <- dagitty( "dag { crc -> microbiome crc <- age -> microbiome crc <- gender -> microbiome }") ggdag(v_dag, text_col="red")+theme_dag()+theme_dag_gray()
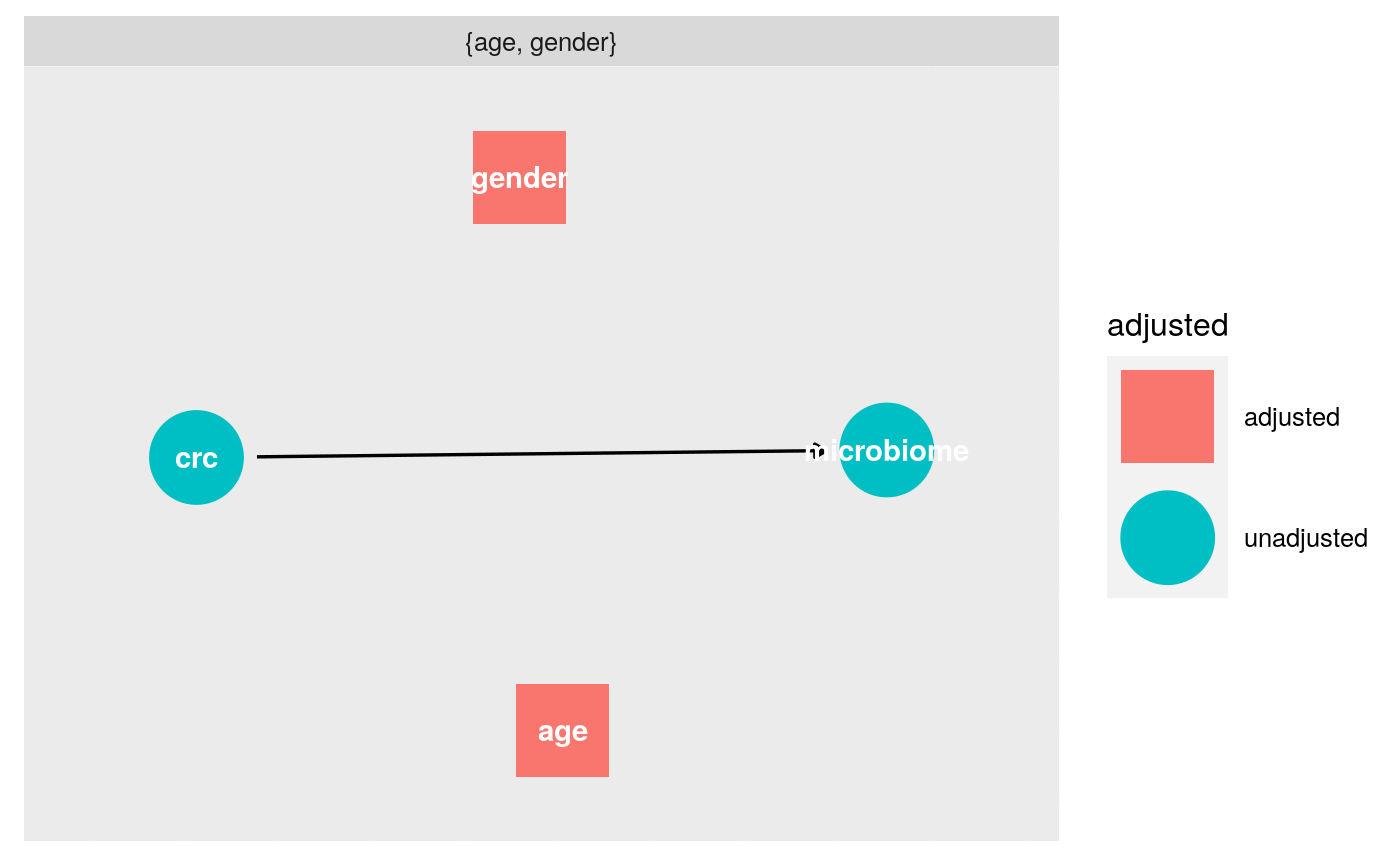
What do we need to adjust for?
ggdag_adjustment_set(v_dag, exposure = "crc", outcome = "microbiome")+theme_dag_gray()
Download the data, extract the expressionset, and only take responses for which we have a value for study condition.
vogtmann <- curatedMetagenomicData("VogtmannE_2016.metaphlan_bugs_list.stool", dryrun = FALSE) vm.eset <-vogtmann[[1]] table(vm.eset$study_condition, useNA="ifany") vm.sub <- vm.eset[,!is.na(vm.eset$study_condition)]
table(vm.sub$study_condition, useNA="ifany")
##
## control CRC
## 52 52First we conduct a basic analysis to examine whether there are any statistically significant differences in taxa of microbiota by study condition. We use a forgiving alpha of 0.10 for adjusted p-values. We find 5 significant taxa in this crude analysis.
vm.pseq <- ExpressionSet2phyloseq(vm.sub) dx_only <- phyloseq_to_deseq2(vm.pseq, ~ study_condition) dx_only <- DESeq(dx_only, test="Wald", fitType="parametric") res <- results(dx_only, cooksCutoff = FALSE) alpha <- 0.10 sigtab <- res[which(res$padj < alpha), ] sigtab <- cbind(as(sigtab, "data.frame"), as(tax_table(vm.pseq)[rownames(sigtab), ], "matrix"))
sigtab## baseMean log2FoldChange lfcSE stat
## g__Escherichia 1.160319 3.131873 0.8680873 3.607785
## s__Ruminococcus_torques 2.057123 1.221872 0.3181531 3.840515
## s__Escherichia_coli 1.021863 3.114437 0.8976217 3.469654
## t__Ruminococcus_torques_unclassified 1.317457 1.711670 0.4640449 3.688588
## t__Escherichia_coli_unclassified 1.021863 3.114437 0.8976217 3.469654
## pvalue padj Kingdom
## g__Escherichia 0.0003088219 0.05507325 Bacteria
## s__Ruminococcus_torques 0.0001227764 0.05507325 Bacteria
## s__Escherichia_coli 0.0005211292 0.05576082 Bacteria
## t__Ruminococcus_torques_unclassified 0.0002255023 0.05507325 Bacteria
## t__Escherichia_coli_unclassified 0.0005211292 0.05576082 Bacteria
## Phylum Class
## g__Escherichia Proteobacteria Gammaproteobacteria
## s__Ruminococcus_torques Firmicutes Clostridia
## s__Escherichia_coli Proteobacteria Gammaproteobacteria
## t__Ruminococcus_torques_unclassified Firmicutes Clostridia
## t__Escherichia_coli_unclassified Proteobacteria Gammaproteobacteria
## Order Family
## g__Escherichia Enterobacteriales Enterobacteriaceae
## s__Ruminococcus_torques Clostridiales Lachnospiraceae
## s__Escherichia_coli Enterobacteriales Enterobacteriaceae
## t__Ruminococcus_torques_unclassified Clostridiales Lachnospiraceae
## t__Escherichia_coli_unclassified Enterobacteriales Enterobacteriaceae
## Genus Species
## g__Escherichia Escherichia <NA>
## s__Ruminococcus_torques Blautia Ruminococcus_torques
## s__Escherichia_coli Escherichia Escherichia_coli
## t__Ruminococcus_torques_unclassified Blautia Ruminococcus_torques
## t__Escherichia_coli_unclassified Escherichia Escherichia_coli
## Strain
## g__Escherichia <NA>
## s__Ruminococcus_torques <NA>
## s__Escherichia_coli <NA>
## t__Ruminococcus_torques_unclassified Ruminococcus_torques_unclassified
## t__Escherichia_coli_unclassified Escherichia_coli_unclassifiedNext we adjust (deconfound) for categorized age. We find two significant results:
dx_age <- phyloseq_to_deseq2(vm.pseq, ~ study_condition + age_category) dx_age <- DESeq(dx_age, test="Wald", fitType="parametric") res = results(dx_age, cooksCutoff = FALSE, contrast=c("study_condition", "CRC", "control")) alpha = 0.10 sigtab = res[which(res$padj < alpha), ] sigtab = cbind(as(sigtab, "data.frame"), as(tax_table(vm.pseq)[rownames(sigtab), ], "matrix"))
sigtab## baseMean log2FoldChange lfcSE stat
## s__Ruminococcus_torques 2.057123 1.225518 0.3199339 3.830534
## t__Ruminococcus_torques_unclassified 1.317457 1.706224 0.4661006 3.660634
## pvalue padj Kingdom
## s__Ruminococcus_torques 0.0001278654 0.06730097 Bacteria
## t__Ruminococcus_torques_unclassified 0.0002515924 0.06730097 Bacteria
## Phylum Class Order
## s__Ruminococcus_torques Firmicutes Clostridia Clostridiales
## t__Ruminococcus_torques_unclassified Firmicutes Clostridia Clostridiales
## Family Genus
## s__Ruminococcus_torques Lachnospiraceae Blautia
## t__Ruminococcus_torques_unclassified Lachnospiraceae Blautia
## Species
## s__Ruminococcus_torques Ruminococcus_torques
## t__Ruminococcus_torques_unclassified Ruminococcus_torques
## Strain
## s__Ruminococcus_torques <NA>
## t__Ruminococcus_torques_unclassified Ruminococcus_torques_unclassifiedFinally we add in gender. Again observing two significant results.
dx_age_gender <- phyloseq_to_deseq2(vm.pseq, ~ study_condition + age_category + gender) dx_age_gender <- DESeq(dx_age_gender, test="Wald", fitType="parametric") res = results(dx_age_gender, cooksCutoff = FALSE, contrast=c("study_condition", "CRC", "control")) alpha = 0.10 sigtab = res[which(res$padj < alpha), ] sigtab = cbind(as(sigtab, "data.frame"), as(tax_table(vm.pseq)[rownames(sigtab), ], "matrix"))
sigtab## baseMean log2FoldChange lfcSE stat
## s__Ruminococcus_torques 2.057123 1.22658 0.3202308 3.830298
## t__Ruminococcus_torques_unclassified 1.317457 1.73073 0.4655552 3.717562
## pvalue padj Kingdom
## s__Ruminococcus_torques 0.0001279881 0.05380886 Bacteria
## t__Ruminococcus_torques_unclassified 0.0002011546 0.05380886 Bacteria
## Phylum Class Order
## s__Ruminococcus_torques Firmicutes Clostridia Clostridiales
## t__Ruminococcus_torques_unclassified Firmicutes Clostridia Clostridiales
## Family Genus
## s__Ruminococcus_torques Lachnospiraceae Blautia
## t__Ruminococcus_torques_unclassified Lachnospiraceae Blautia
## Species
## s__Ruminococcus_torques Ruminococcus_torques
## t__Ruminococcus_torques_unclassified Ruminococcus_torques
## Strain
## s__Ruminococcus_torques <NA>
## t__Ruminococcus_torques_unclassified Ruminococcus_torques_unclassifiedAs mentioned in the confounding section, estimates obtained for the relationship between the exposure and the outcome have been adjusted for the included confounders (deconfounded). This means that the observed estimate is an averaged effect size across strata of the confounder(s).
Toy Problems
- Consider the DAG below. What variables should you control for if you want to measure the effect of X on Y?

- Create your own DAG (of a causal relationship of interest to you) then identify what variables you would need to control for to measure the causal effect of interest.
- One important subject that I did not touch on in this workshop was generalizability or transportability of causal findings (applying the results to an external population). What issues might arise when doing so? How can a DAG be used to resolve issues of generalizability and transportability?
Conclusions
Unfortunately there’s no silver bullet for confounding or bias in R or Bioconductor. Instead, we as researchers must carefully consider possible sources of bias–ideally as we begin designing the study. DAGs are helpful in providing a clear visualization of the hypothesized causal mechanism and identifying potential confounders. As Hernan says, “Draw your assumptions before you draw your conclusions.”
Unmeasured confounding presents a particularly difficult problem for causal inference. Because the confounder was not measured, we cannot easily deconfound for it.
It is important for us to remember that the definition of a cause is rooted in the counterfactual: we need to find a way to come as close as possible to observing what would have happened if a participant’s exposure status had been different.
Further Reading/Materials
This has been a basic primer on causal inference but if you’d like to learn more:
- Pearl and Mackenzie Book of Why: The New Science of Causal Effect (easy summer beach read!)
- Hernan and Robins Causal Inference available free online: https://www.hsph.harvard.edu/miguel-hernan/causal-inference-book/
- Pearl Causality: Models, Reasoning, and Inference
- Hernan’s edX course available free online: https://www.edx.org/course/causal-diagrams-draw-your-assumptions-before-your